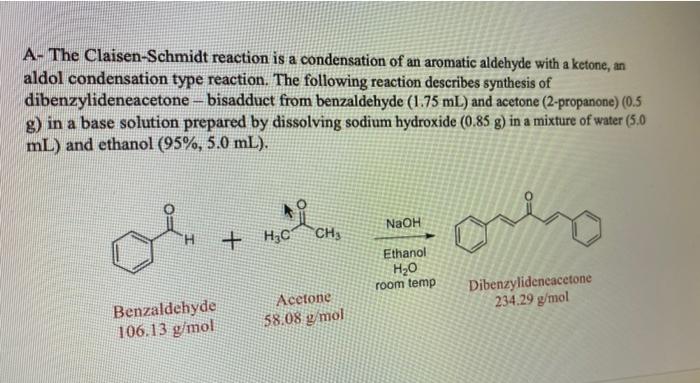
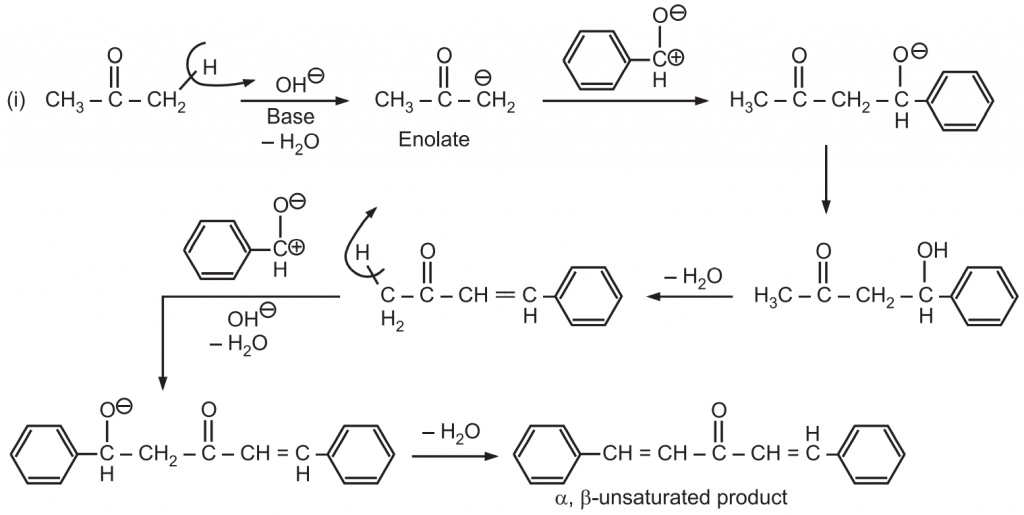
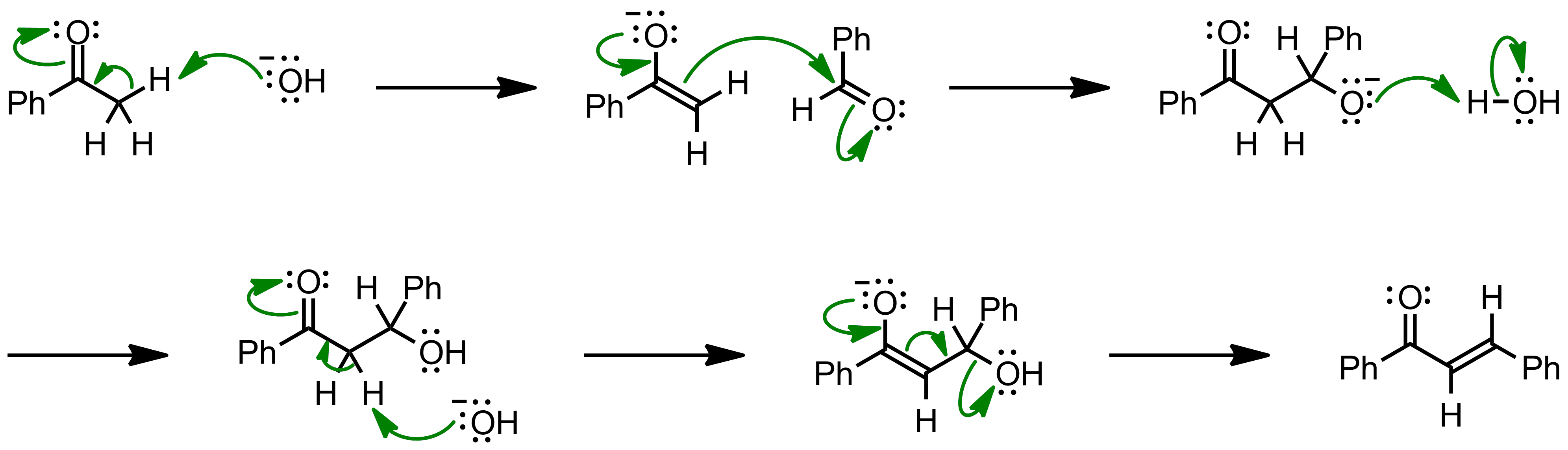
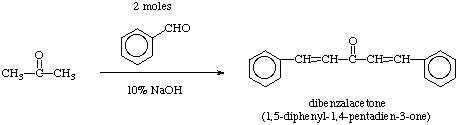
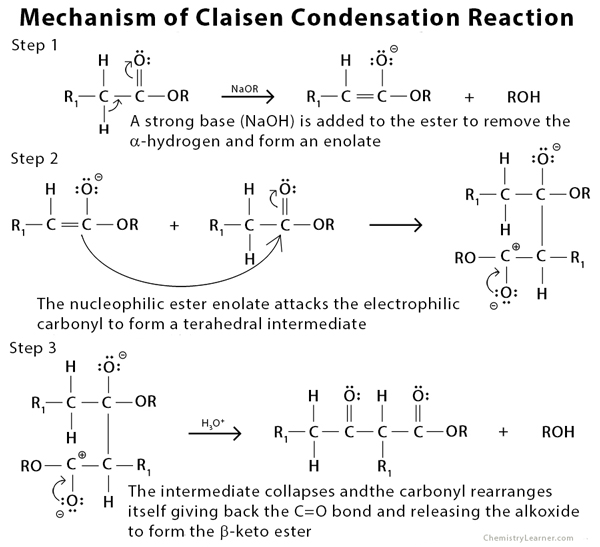
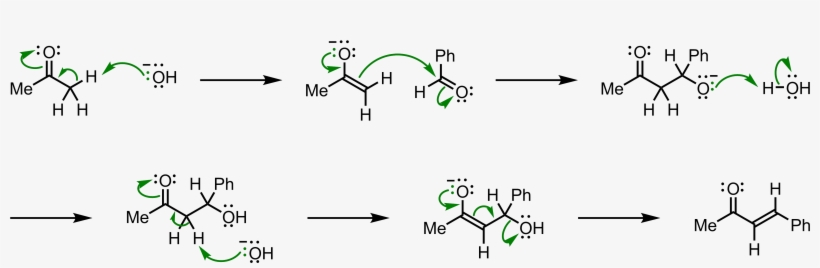
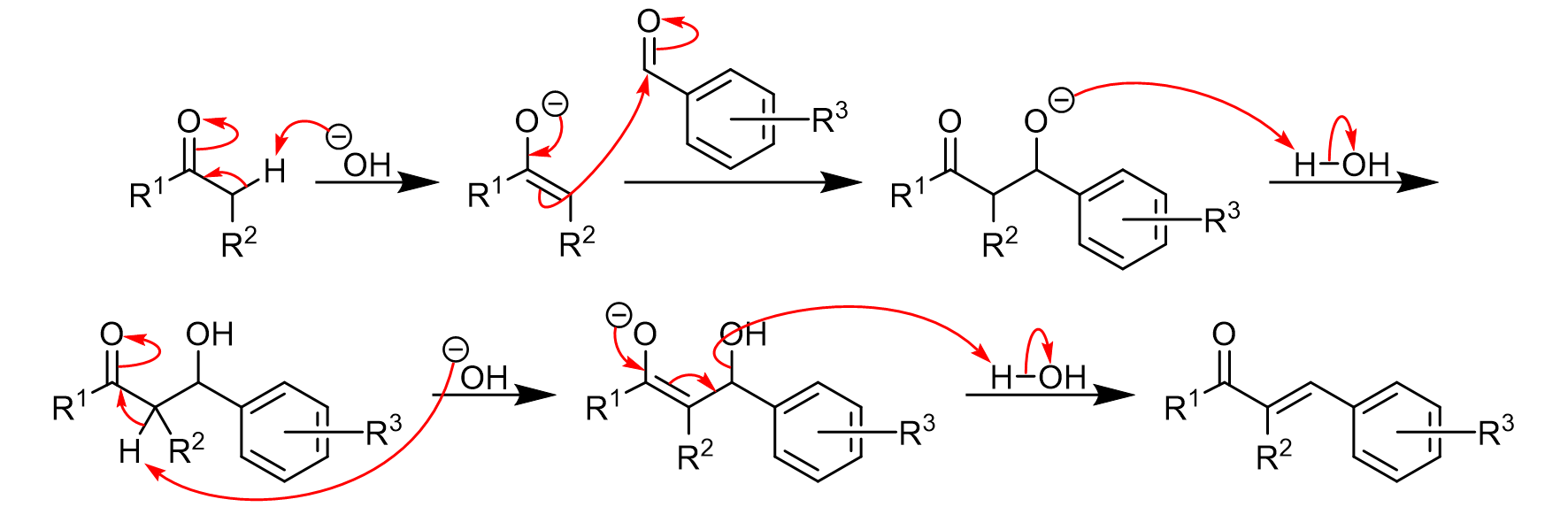
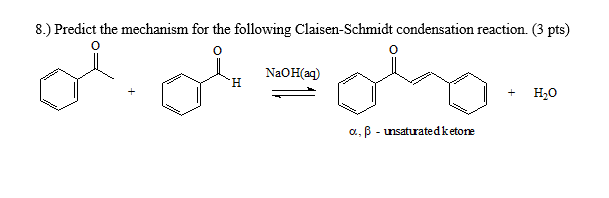
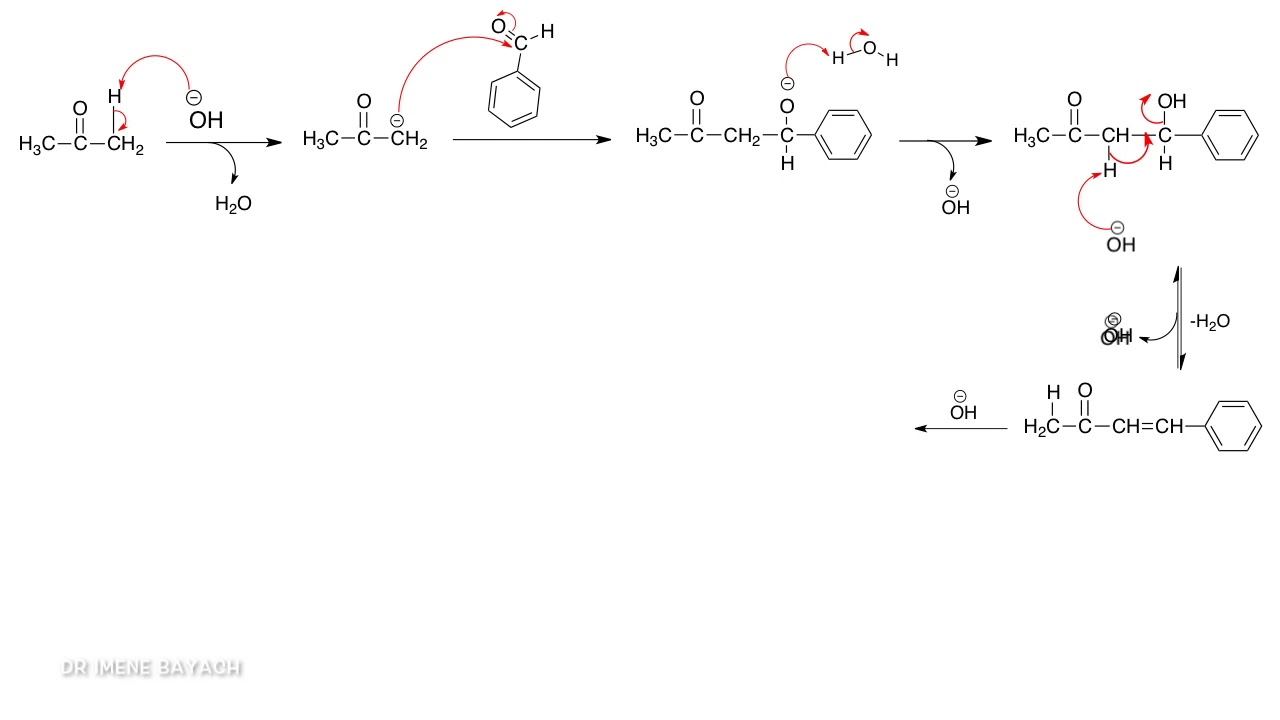
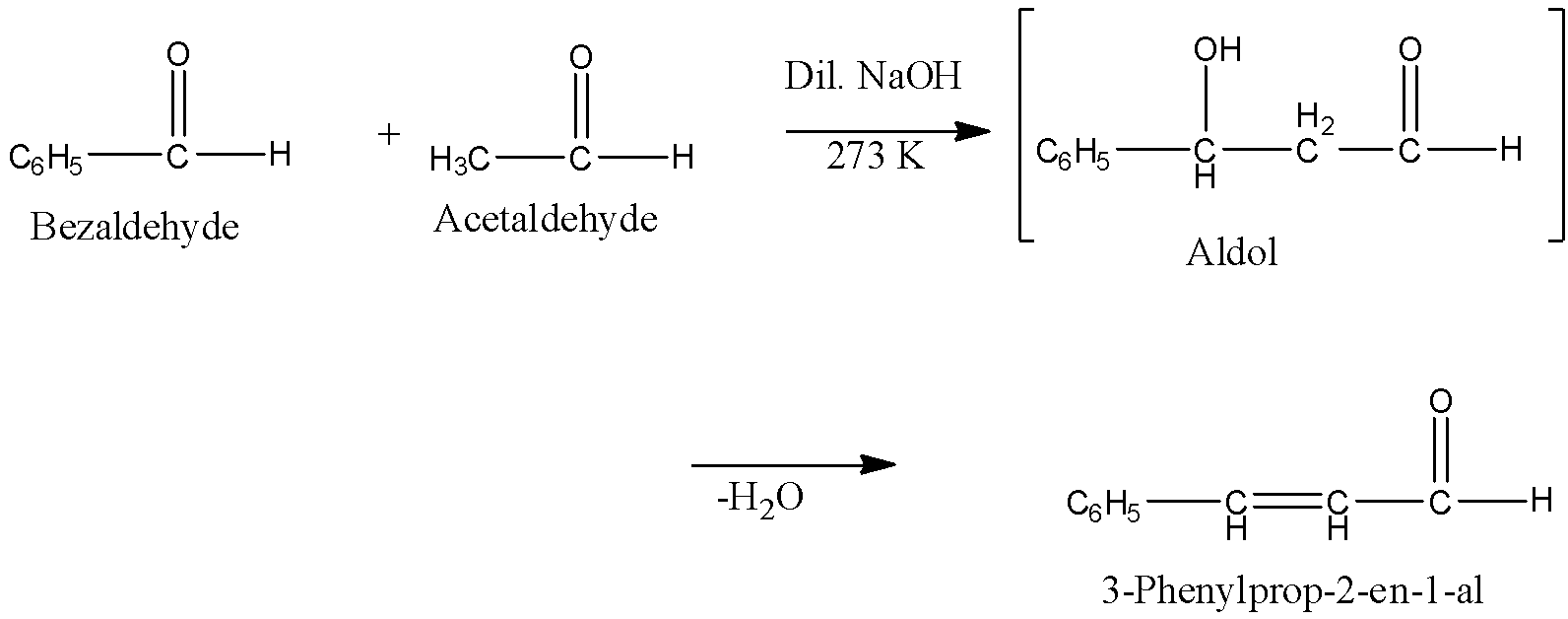
Why Benzaldehyde and acetone can't do self-condensation in claisen-Schmidt reaction condensation? 2)In the second step of the same reaction, why do we add ethanol to form dibenzalideneacetone? | Homework.Study.com
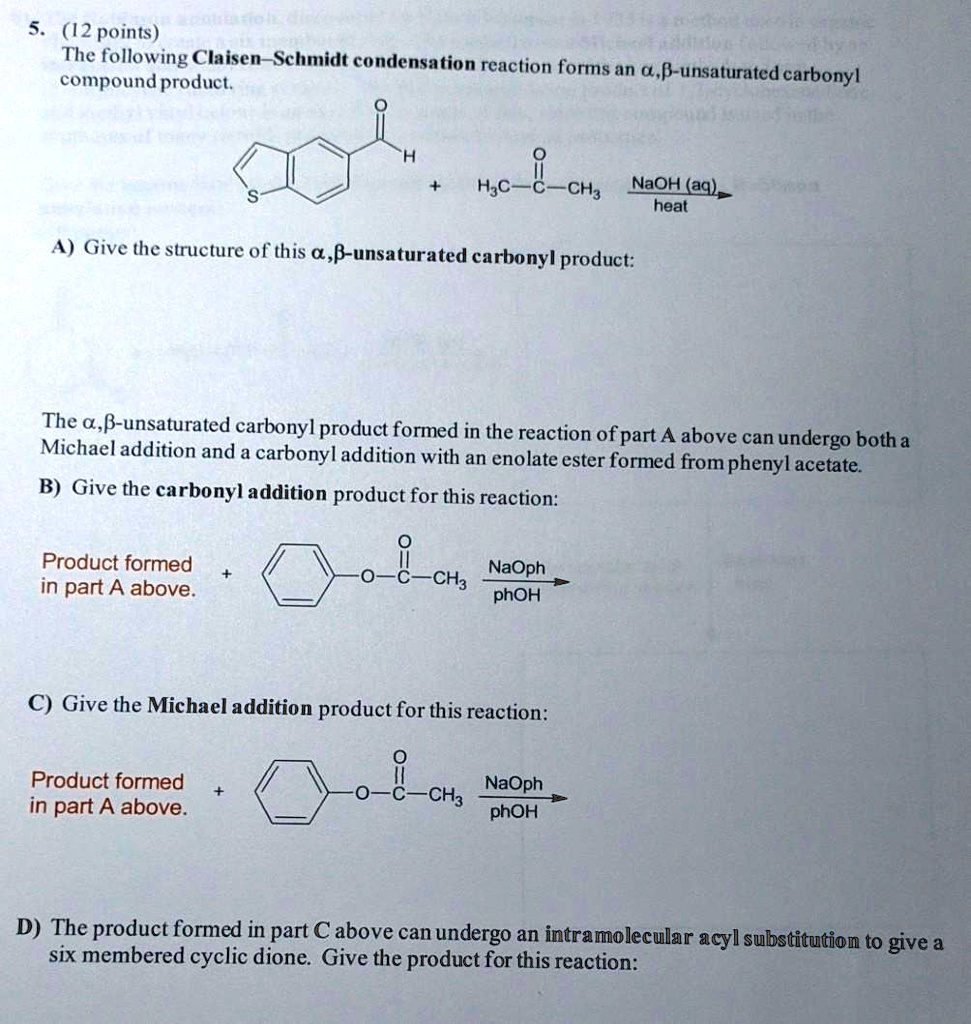
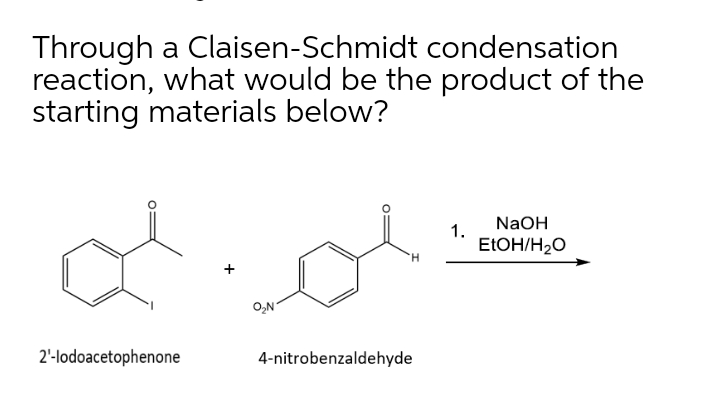
SOLVED: (12 points) The following Claisen-Schmidt condensation reaction forms an G,f-unsaturated e compound product; Icarbonyl H;c CH3 NaQH(aq) heat A) Give the structure Of this a,p-unsaturated carbonyl product: The ,B-unsaturated carbonyl product

An efficient and selective microwave-assisted Claisen-Schmidt reaction for the synthesis of functionalized benzalacetones | SpringerPlus | Full Text